Lithium Chromate
Lithium Chromate Trade Information
- Supply Ability
- 100 Per Month
- Delivery Time
- 7 Days
About Lithium Chromate
| Name of Product | Lithium Chromate | IUPAC Name | dilithium;dioxido(dioxo)chromium |
| CAS No | 14307-35-8 | InChI | InChI=1S/Cr.2Li.4O/q;2*+1;;;2*-1 |
| Formula | CrLi2O4 | InChI Key | JQVALDCWTQRVQE-UHFFFAOYSA-N |
| Other name / synonyms | LITHIUM CHROMATE | Canonical SMILES | [Li+].[Li+].[O-][Cr](=O)(=O)[O-] |
| Dilithium chromate | EC Number | 238-244-7 | |
| Chromium lithium oxide | UN Number | 3077 | |
| Lithium chromate(VI) | UNII | 5UR77B9Q8M | |
| Chromic acid, dilithium salt | |||
| Lithium chromate (Li2CrO4) | |||
| Dilithium chromate (Li2CrO4) | |||
| HSDB 650 | |||
| 14307-35-8 | |||
| Chromium lithium oxide (CrLi2O4) | |||
| Lithium chromium oxide (Li2CrO4) | |||
| EINECS 238-244-7 | |||
| Chromic acid (H2CrO4), dilithium salt | |||
| dilithium dioxido(dioxo)chromium | |||
| Lithiumchromate | |||
| AC1L1BJF | |||
| AC1MTPF1 | |||
| UNII-5UR77B9Q8M | |||
| 5UR77B9Q8M | |||
| CTK8B0780 | |||
| ANW-20736 | |||
| AKOS015904488 | |||
| LS-53390 | |||
| S426 | |||
| dilithium(1+) ion dioxochromiumbis(olate) | |||
| FT-0694957 | |||
| Chromic acid (H2CrO4), lithium salt (1:2) | |||
| 3B4-4228 | |||
| I14-17283 |
Key Structural Features
Lithium Chromate features the chromate ion (CrO4), where a chromium atom is centrally located within a tetrahedral arrangement of oxygen atoms. Its structural integrity supports diverse chemical interactions, making it valuable in laboratory synthesis and industrial processes that rely on chromate chemistry.
Industrial Applications
While specific uses are not detailed, compounds containing chromate ions like Lithium Chromate are frequently utilized for dyeing, pigmentation, corrosion inhibition, and analytical chemistry. Its unique molecular structure allows for specialized use in sectors requiring reliable chromate sourcing from reputable Indian exporters and suppliers.
FAQ's of Lithium Chromate:
Q: How is Lithium Chromate typically used in industry?
A: Though exact applications are not specified, Lithium Chromate as a compound containing chromate ions is often employed in the production of pigments, corrosion inhibitors, and laboratory reagents, taking advantage of the reactivity of the CrO4 ion.Q: What benefits does Lithium Chromate provide in chemical processes?
A: Lithium Chromate offers the advantage of stable chromate ion supply, essential for reactions that require oxidation or synthesis capacities associated with chromate-based compounds.Q: When should Lithium Chromate be handled with precaution?
A: Handling Lithium Chromate demands caution at all times, as compounds containing chromate ions are typically considered hazardous and require appropriate protective measures and regulatory compliance.Q: Where can Lithium Chromate be sourced from in India?
A: Lithium Chromate can be obtained through various chemical manufacturers, exporters, and suppliers across India, who provide the compound for laboratory and industrial needs.Q: What is the process for purchasing Lithium Chromate from Indian suppliers?
A: Clients can approach verified Indian exporters or manufacturers directly, generally through online B2B platforms or specialized chemical trading sites, to request specifications, pricing, and secure procurement.Q: How does the structural formula of Lithium Chromate contribute to its chemical behavior?
A: The CrO4 structure imparts significant oxidative properties, making the compound suitable for specialized synthetic and analytical functions where chromate ions are required.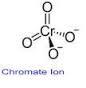

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Lithium Compounds Category
Lithium Bromide
Solubility : Easily soluble in water, ethanol, and ether
Molecular Formula : LiBr
Usage : Used in absorption chillers, industrial drying, air conditioning, and pharmaceuticals
Grade : Other, Industrial Grade / Laboratory Grade
Molecular Weight : 86.85 g/mol
Storage : Other, Store in tightly closed container, dry and cool place
Lithium Sulphate Li2SO4
Solubility : Soluble in water (29 g/100 ml at 20C); insoluble in alcohol
Molecular Formula : Li2SO4
Usage : Industrial and laboratory
Grade : Other, Analytical/Laboratory grade
Molecular Weight : 109.94 g/mol
Storage : Other, Store in a cool, dry and wellventilated place
Anhydrous Lithium Bromide Powder
Solubility : Highly soluble in water, alcohol, and ether
Molecular Formula : LiBr
Usage : Industrial and laboratory
Grade : Industrial Grade
Molecular Weight : 86.85 g/mol
Storage : Other, Store in tightly closed container, in a cool, dry, and wellventilated place
Lithium Chromate Solution 20%
Solubility : Soluble in water
Molecular Formula : Li2CrO4 (for pure substance)
Usage : Laboratory chemical, industrial applications
Grade : Other, Laboratory/Technical Grade
Molecular Weight : 117.91 g/mol (for pure substance)
Storage : Other, Store in cool, dry, wellventilated area; tightly closed container

 Send Inquiry
Send Inquiry




 Send Inquiry
Send Inquiry Send SMS
Send SMS
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese