Bromobenzene
Bromobenzene Specification
- Smell
- Benzene-like odor
- Refractive Rate
- 1.561
- Molecular Formula
- C6H5Br
- Molecular Weight
- 157.01 g/mol
- Structural Formula
- Br-C6H5
- HS Code
- 29036990
- Boiling point
- 156 C
- Classification
- Aromatic halogen compound
- Grade
- Industrial
- Purity
- 99%
- Application
- Used as intermediates in organic synthesis
- Appearance
- Transparent liquid
- CAS No
- 108-86-1
- EINECS No
- 203-623-8
- Other Names
- Phenyl bromide
- Usage
- Chemical intermediate; solvent
- Melting Point
- -30 C
- Density
- 1.495 Gram per cubic centimeter(g/cm3)
- Solubility
- Insoluble in water soluble in organic solvents
- Raw Material
- Benzene
Bromobenzene Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 100 Kilograms Per Month
- Delivery Time
- 7 Days
About Bromobenzene
| Structure : C6H5Br |
| Molecular formula : C6H5Br |
| Molecular weight : 157.02 |
| Chemical name : Bromobenzene |
Synonyms
1-Bromobenzene;
Benzene,bromo-;
Brombenzol;
bromo-benzen;
Bromobenzol;
NCI-C55492;
PHENYL BROMIDE;
MONOBROMOBENZENE
Key Properties and Chemical Composition
Bromobenzene features a transparent appearance, high purity (99%), and a density of 1.495 g/cm3. Its molecular structure consists of a bromine atom attached to a benzene ring, making it ideal for a range of chemical processes. The product is recognized by its CAS No: 108-86-1 and EINECS No: 203-623-8.
Applications in Organic Synthesis
Bromobenzene serves as a crucial intermediate in the synthesis of various organic compounds. It is predominantly utilized in the pharmaceutical, agrochemical, and dye industries for its effective chemical properties. Additionally, bromobenzene acts as a solvent in specialized laboratory reactions.
Handling, Storage, and Export Details
The product should be stored in tightly sealed containers away from moisture and direct sunlight to maintain its quality. Primarily exported and supplied from India, bromobenzene complies with relevant industry standards and is shipped by qualified manufacturers and suppliers.
FAQ's of Bromobenzene:
Q: How is bromobenzene commonly used in industrial applications?
A: Bromobenzene is primarily utilized as a chemical intermediate in organic synthesis, particularly in the production of pharmaceuticals, agrochemicals, and dyes. It is also used as a solvent in laboratory settings where its unique chemical properties are advantageous.Q: What is the process for manufacturing bromobenzene?
A: Bromobenzene is manufactured via the bromination of benzene, where benzene reacts with bromine in the presence of an iron catalyst. This controlled reaction yields high-purity bromobenzene suitable for industrial and laboratory use.Q: When should bromobenzene be used as a solvent rather than another compound?
A: Bromobenzene should be chosen as a solvent when working with organic compounds that require a nonpolar or weakly polar medium and where solubility in water is not desired. Its benzene-like characteristics make it particularly valuable in Grignard and coupling reactions.Q: Where is bromobenzene typically stored and how should it be handled safely?
A: Bromobenzene should be stored in well-ventilated, cool, and dry locations within tightly closed containers. Proper safety measures, such as using gloves and eye protection, are recommended when handling to avoid skin and eye contact due to its volatile nature.Q: What are the main benefits of using bromobenzene as a chemical intermediate?
A: The principal benefits include its high reactivity for producing more complex aromatic compounds, reliable performance in various organic reactions, and compatibility with standard laboratory and industrial processes.Q: Is bromobenzene soluble in water and what solvents is it compatible with?
A: Bromobenzene is insoluble in water but dissolves readily in many organic solvents such as ether, chloroform, and alcohol. Its solubility profile makes it versatile for different organic synthesis applications.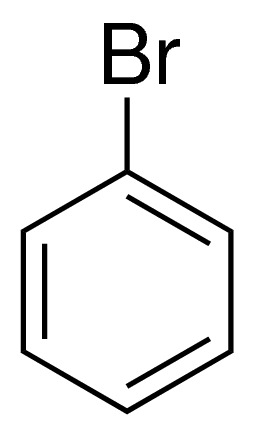

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Aliphatic Bromide Category
n-Propyl Acetate
Minimum Order Quantity : 1 Kilograms
Purity : 99.5%
Other Names : 1Propyl acetate; Propyl ethanoate
Structural Formula : CH3COOCH2CH2CH3
Raw Material : Propyl alcohol, Acetic acid
HS Code : 2915399090
Potassium Bromide
Minimum Order Quantity : 1 Kilograms
Purity : 99% min
Other Names : KBr
Structural Formula : K+ Br
Raw Material : Potassium Carbonate, Hydrobromic Acid
HS Code : 28275120
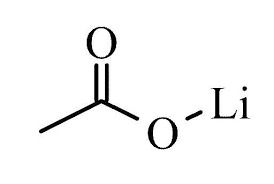
Lithium Acetate Dihydrate
Minimum Order Quantity : 1 Kilograms
Purity : 99%
Other Names : Lithium acetate hydrate
Structural Formula : LiC2H3O22H2O
Raw Material : Acetic acid and lithium compounds
HS Code : 2915.32.00
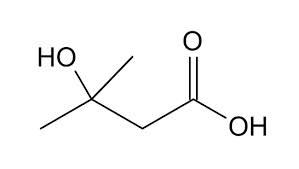
Methyl 3 -Oxopentanoate
Minimum Order Quantity : 1 Kilograms
Purity : 98%
Other Names : Methyl acetylpropanoate
Structural Formula : HOC(CH3)CH2C(=O)OCH3
Raw Material : Derived from acetoacetic acid and methanol
HS Code : 29183000

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese