Methyl Chloro Acetate
Methyl Chloro Acetate Specification
- Smell
- Pleasant fruity odor
- Molecular Weight
- 108.52 g/mol
- Refractive Rate
- 1.421 (20C)
- Molecular Formula
- C3H5ClO2
- Structural Formula
- Cl-CH2-COOCH3
- HS Code
- 2915
- Boiling point
- 107.5C
- Classification
- Organic compound
- Organic Acid Types
- Halogenated ester
- Grade
- Industrial Grade
- Purity
- 98%
- Application
- Used as an intermediate in organic synthesis
- Appearance
- Colorless liquid
- CAS No
- 96-34-4
- EINECS No
- 202-504-8
- Other Names
- Chloroacetic acid methyl ester; Methyl 2-chloroacetate
- Usage
- Used in pharmaceuticals agrochemicals and dyes production
- Melting Point
- -45C
- Density
- 1.231 Gram per cubic centimeter(g/cm3)
- Solubility
- Slightly soluble in water soluble in organic solvents
- Raw Material
- Chloroacetic acid or derivatives
Methyl Chloro Acetate Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 100 Kilograms Per Month
- Delivery Time
- 7 Days
About Methyl Chloro Acetate
| Name of Product | Methyl Chloro Acetate | IUPAC Name | methyl 2-chloroacetate |
| CAS No | 96-34-4 | InChI | InChI=1S/C3H5ClO2/c1-6-3(5)2-4/h2H2,1H3 |
| Formula | C3H4BrClO2 | InChI Key | QABLOFMHHSOFRJ-UHFFFAOYSA-N |
| Other name / synonyms | Chloromethyl acetate | Canonical SMILES | COC(=O)CCl |
| 625-56-9 | CAS | 96-34-4 | |
| Chloromethyl ethanoate | EC Number | 202-501-1 | |
| Methanol, chloro-, acetate | ICSC Number | 1410 | |
| SMJYMSAPPGLBAR-UHFFFAOYSA-N | RTECS Number | AF9500000 | |
| Chloromethanol acetate | UN Number | 2295 | |
| Chloromethyl acetate # | UNII | 450VSB182I | |
| AC1L2BPH | |||
| AC1Q3VDL | Description | Clear, colourless liquid | |
| ghl.PD_Mitscher_leg0.478 | Acidity | Not more than 0.5 % | |
| acetic acid chloromethyl ester | Moisture (KF) | Not more than 0.1 % | |
| KSC492I3P | Purity by GC | Not less than 98.0% | |
| SCHEMBL194595 | |||
| CTK3J2437 | |||
| MolPort-001-785-628 | |||
| EBD31608 | |||
| EINECS 210-902-8 | |||
| ANW-34264 | |||
| AR-1I2057 | |||
| AKOS006274758 | |||
| RTC-030591 | |||
| AN-46828 | |||
| CJ-30179 | |||
| SC-47539 | |||
| AB0052938 | |||
| TC-030591 | |||
| FT-0080621 | |||
| FT-0600220 | |||
| Y1150 | |||
| A24540 | |||
| Chloromethyl ethanoate;Acetic acid chloromethyl | |||
| S-7939 | |||
| I04-0268 | |||
| 3B3-067155 |
A Versatile Intermediate for Organic Synthesis
Methyl Chloro Acetate serves as a critical building block in the synthesis of various organic compounds. Its reactivity as a halogenated ester enables the efficient production of pharmaceuticals, agrochemicals, and dyes. With a high level of purity and consistent quality, this substance supports complex chemical processes across industries, making it an essential component in modern manufacturing.
Physical Properties and Handling Requirements
This compound appears as a colorless liquid with a distinctive fruity smell, making its identification straightforward. Its melting point is -45C and it boils at 107.5C. Methyl Chloro Acetate is slightly soluble in water but is easily soluble in many organic solvents. Proper storage in well-ventilated and temperature-controlled areas is recommended to maintain product integrity for industrial applications.
FAQ's of Methyl Chloro Acetate:
Q: How is Methyl Chloro Acetate manufactured?
A: Methyl Chloro Acetate is typically synthesized using chloroacetic acid or its derivatives as raw materials, often via esterification processes involving methanol. Specialized equipment is required to handle its chemical reactivity and ensure high purity for industrial use.Q: What are the primary industrial applications of Methyl Chloro Acetate?
A: This compound is mainly used as an intermediate in the production of pharmaceuticals, agrochemicals, and dyes. Its chemical structure allows it to participate in a variety of organic reactions essential for synthesizing complex molecules.Q: When should Methyl Chloro Acetate be used in a chemical process?
A: Methyl Chloro Acetate is best used when a reactive halogenated ester is needed to introduce chloroacetyl groups or facilitate organic transformations in the manufacture of specialty chemicals and bioactive compounds.Q: Where is Methyl Chloro Acetate produced and supplied from?
A: Manufacturers and exporters in India are notable suppliers of Methyl Chloro Acetate, providing both domestic and international markets with industrial-grade material that meets quality and purity standards.Q: What is the benefit of using Methyl Chloro Acetate in synthesis?
A: Due to its high reactivity and purity (98%), Methyl Chloro Acetate ensures efficient yields and selective reactions, which are vital in the multi-step preparation of pharmaceuticals and other fine chemicals.Q: How should Methyl Chloro Acetate be stored and handled safely?
A: It should be stored in tightly sealed containers in a cool, well-ventilated area away from incompatible materials. Appropriate personal protective equipment is recommended when handling to avoid exposure.Q: Is Methyl Chloro Acetate soluble in water or organic solvents?
A: Methyl Chloro Acetate is slightly soluble in water but is easily soluble in a wide range of organic solvents, which makes it convenient for use in diverse chemical synthesis processes.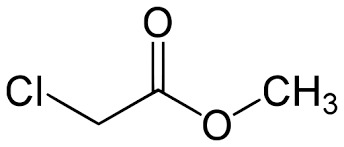

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Aliphatic Bromide Category
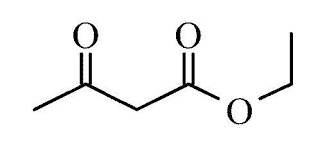
Ethyl Aceto Acetate
Minimum Order Quantity : 1 Kilograms
Appearance : Clear, colourless liquid
HS Code : 29183000
Raw Material : Acetic acid, Ethanol
Structural Formula : CH3COCH2COOC2H5
EINECS No : 2055161
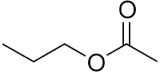
N-Propyl Acetate
Minimum Order Quantity : 1 Kilograms
Appearance : Colorless liquid
HS Code : 2915.39
Raw Material : Propanol Acetic acid
Structural Formula : CH3COOCH2CH2CH3
EINECS No : 2036861
N Pentyl Bromide
Minimum Order Quantity : 1 Kilograms
Appearance : Clear colorless liquid
HS Code : 2903 39 90
Raw Material : Pentanol
Structural Formula : CH3(CH2)4Br
EINECS No : 2038574
Bromoform
Minimum Order Quantity : 1 Kilograms
Appearance : Clear yellow liquid
HS Code : 2903.11
Raw Material : Bromine
Structural Formula : CHBr
EINECS No : 2008567

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese