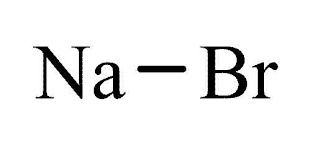About सà¥à¤¡à¤¿à¤¯à¤® बà¥à¤°à¥à¤®à¤¾à¤à¤¡
| Name of Product | SODIUM BROMIDE | IUPAC Name | sodium;bromide |
| CAS No | 7647-15-6 | InChI | InChI=1S/BrH.Na/h1H;/q;+1/p-1 |
| Formula | NaBr | InChI Key | JHJLBTNAGRQEKS-UHFFFAOYSA-M |
| Other name / synonyms | SODIUM BROMIDE | Canonical SMILES | [Na+].[Br-] |
| Bromide salt of sodium | CAS | 7647-15-6 |
| 7647-15-6 | EC Number | 231-599-9 |
| Sodium bromide (NaBr) | UNII | LC1V549NOM |
| Sedoneural | Wikipedia | Sodium bromide |
| NaBr |
|
|
| Trisodium tribromide | Description | White grannular powder |
| Bromnatrium | Loss on drying | Not more than 1.0 % |
| Bromnatrium [German] | Assay (on dried basis) | NLT 98.5 % |
| Caswell No. 750A |
|
|
| UNII-LC1V549NOM |
|
|
| CHEMBL1644694 |
|
|
| CHEBI:63004 |
|
|
| HSDB 5039 |
|
|
| EINECS 231-599-9 |
|
|
| NSC 77384 |
|
|
| EPA Pesticide Chemical Code 013907 |
|
|
| 59217-63-9 |
|
|
| bromosodium |
|
|
| Sodiumbromide |
|
|
| Sodium bromide (TN) |
|
|
| Sodium bromide solution |
|
|
| Sodium bromide [JAN] |
|
|
| ACMC-1BDFJ |
|
|
| Bromide standard for IC |
|
|
| AC1Q1UKI |
|
|
| Sodium bromide (JP16) |
|
|
| WLN: NA E |
|
|
| SODIUM BROMIDE, ACS |
|
|
| AC1L5P1N |
|
|
| AC1Q1UK0 |
|
|
| DSSTox_CID_14903 |
|
|
| DSSTox_RID_79219 |
|
|
| LC1V549NOM |
|
|
| DSSTox_GSID_34903 |
|
|
| KSC377C0R |
|
|
| 02119_RIEDEL |
|
|
| 229881_ALDRICH |
|
|
| 460591_ALDRICH |
|
|
| S4547_SIAL |
|
|
| 01963_FLUKA |
|
|
| 17355_FLUKA |
|
|
| 71329_FLUKA |
|
|
| 71329_SIGMA |
|
|
| CTK2H7108 |
|
|
| JHJLBTNAGRQEKS-UHFFFAOYSA-M |
|
|
| MolPort-003-925-189 |
|
|
| MolPort-003-987-220 |
|
|
| 02119_SIAL |
|
|
| 220345_SIAL |
|
|
| 310506_SIAL |
|
|
| NSC77384 |
|
|
| Tox21_301343 |
|
|
| NSC-77384 |
|
|
| AKOS024438090 |
|
|
| RP18797 |
|
|
| RTR-024487 |
|
|
| TRA-0176241 |
|
|
| NCGC00255632-01 |
|
|
| AN-21811 |
|
|
| CAS-7647-15-6 |
|
|
| LS-145526 |
|
|
| TL8005224 |
|
|
| TR-024487 |
|
|
| FT-0645125 |
|
|
| S0546 |
|
|
| D02055 |
|
|
| 3B4-1883 |
|
|
| I14-92078 |
|
|
Product Quality and StabilitySodium Bromide ensures consistent quality with a strict moisture content of 0.5% and a low level of heavy metals (5 ppm). Its stability under normal conditions means it retains its efficacy when stored correctly, minimizing the risk of degradation or contamination.
Versatile ApplicationsWith applications ranging from industrial processing to laboratory analysis and photographic development, Sodium Bromide offers unparalleled flexibility. It is particularly valued in drilling fluids, water treatment, and as a sedative precursor in pharmaceutical formulations.
Safe Handling and StorageBest stored in cool, dry, and well-ventilated environments away from humidity, Sodium Bromide is supplied in robust HDPE bags with an inner liner to protect from moisture. Detailed packaging options support a variety of usage scales and customer requirements.
FAQ's of Sodium Bromide :
Q: How should Sodium Bromide be stored to maintain its stability and purity?
A: To maintain the stability and high purity of Sodium Bromide, store it in a cool, dry, and well-ventilated area, away from humidity. Use the original HDPE packaging with inner liner to prevent moisture absorption and contamination.
Q: What are the key industrial and laboratory uses of Sodium Bromide?
A: Sodium Bromide is widely used as a source of bromide ions for preparing other bromides, in photographic processes, drilling fluids, water treatment, and as an analytical reagent in laboratories. It also figures in the pharmaceutical sector as a sedative precursor.
Q: When is Sodium Bromide typically applied in drilling operations?
A: Sodium Bromide is added to drilling fluids during oil and gas exploration to stabilize boreholes, control formation pressures, and prevent well collapse, leveraging its high solubility and chemical stability.
Q: Where is Sodium Bromide manufactured and exported from?
A: Produced using high-quality raw materials such as bromine, sodium carbonate, and sodium hydroxide, Sodium Bromide is manufactured and supplied by leading exporters based in India to global markets.
Q: What processes are involved in the production of Sodium Bromide?
A: The production of Sodium Bromide involves the reaction of bromine with sodium carbonate or sodium hydroxide, followed by purification steps to achieve high purity (99%) and low moisture and chloride contents.
Q: How does Sodium Bromide benefit water treatment systems?
A: Sodium Bromide acts as an efficient source of bromide ions in water treatment systems, enhancing the disinfection process and improving the removal of organic contaminants, all while being highly soluble and easy to handle.
Q: What safety precautions should be observed when handling Sodium Bromide?
A: Ensure appropriate personal protective equipment is worn, avoid direct inhalation or contact with skin or eyes, and always handle the chemical in accordance with recommended storage and safety guidelines.