2-Bromobutane 98%
2-Bromobutane 98% Specification
- HS Code
- 2903.99
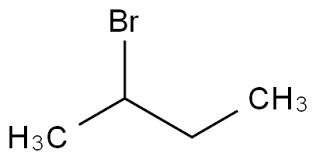
- Molecular Formula
- C4H9Br
- Molecular Weight
- 137.02 g/mol
- Smell
- Sweet ethereal odor
- Refractive Rate
- 1.436
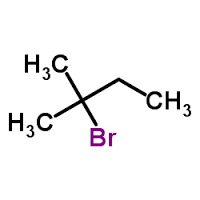
- Structural Formula
- CH3CH(Br)CH2CH3
- Boiling point
- 91C
- Classification
- Halogenated hydrocarbon
- Grade
- Industrial Grade
- Purity
- 98%
- Application
- Organic synthesis
- Appearance
- Colorless liquid
- CAS No
- 78-76-2
- EINECS No
- 201-140-1
- Other Names
- bromo-2-butane
- Usage
- Used as a solvent and intermediate in chemical syntheses
- Melting Point
- -112C
- Density
- 1.22 Gram per cubic centimeter(g/cm3)
- Solubility
- Insoluble in water soluble in ethanol and ether
- Raw Material
- Butane
2-Bromobutane 98% Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 100 Kilograms Per Month
- Delivery Time
- 7 Days
About 2-Bromobutane 98%
| Name of Product | 2-Bromobutane |
| CAS No | 78-76-2 |
| Formula | C4H9Br |
| IUPAC Name | 2-bromobutane |
| InChI | InChI=1S/C4H9Br/c1-3-4(2)5/h4H,3H2,1-2H3 |
| InChI Key | UPSXAPQYNGXVBF-UHFFFAOYSA-N |
| Canonical SMILES | CCC(C)Br |
| EC Number | 201-140-7 |
| UN Number | 2339 |
| Other name / synonyms | 2-Bromobutane |
| SEC-BUTYL BROMIDE | |
| 78-76-2 | |
| Butane, 2-bromo- | |
| 2-Butyl bromide | |
| Methylethylbromomethane | |
| 2-Bromo-butane | |
| sec-Butylbromide | |
| Secondary butyl bromide | |
| 1-Bromo-1-methylpropane | |
| CCRIS 106 | |
| HSDB 2197 | |
| UPSXAPQYNGXVBF-UHFFFAOYSA-N | |
| NSC 8417 | |
| EINECS 201-140-7 | |
| SBB059931 | |
| UN2339 | |
| BRN 0505949 | |
| AI3-25262 | |
| 3-bromobutane | |
| 2-Bromo butane | |
| Butane,2-bromo- | |
| sJPHAbILuP@ | |
| sec-C4H9Br | |
| PubChem3753 | |
| 1-Methylpropyl bromide | |
| S-BUTYL BROMIDE | |
| AC1L1MSD | |
| BROMOBUTANE, 2- | |
| DSSTox_CID_1499 | |
| ACMC-209pf5 | |
Versatile Organic Synthesis Intermediate
2-Bromobutane serves as a valuable building block in organic chemistry. Its reactivity enables the formation of a wide range of compounds, making it an essential reagent in research laboratories and industrial-scale chemical production. Its structure facilitates efficient alkylation and substitution reactions, broadening its utility in synthesizing pharmaceuticals, agrochemicals, and specialty chemicals.
Industrial Strength and Consistent Quality
Sourced from high-quality butane, our 2-Bromobutane is manufactured to meet industrial-grade standards. With a tightly controlled production process, the 98% purity level ensures reliable results in every batch. Our quality assurance measures support consistent performance, which is critical for demanding synthesis procedures in various industrial sectors.
FAQ's of 2-Bromobutane 98%:
Q: How should 2-Bromobutane be safely stored and handled?
A: 2-Bromobutane should be stored in a tightly sealed container in a cool, well-ventilated area away from sources of ignition and strong oxidizers. Protective gear, including gloves and eye protection, is recommended during handling to prevent skin and eye contact. Adequate ventilation helps control vapors due to its volatile nature.Q: What are the primary industrial applications of 2-Bromobutane?
A: 2-Bromobutane is widely used as a solvent and an intermediate in organic synthesis. It plays a crucial role in preparing pharmaceuticals, agrochemicals, and fine chemicals, owing to its reactive bromine atom which facilitates substitution and elimination reactions.Q: When is 2-Bromobutane preferred over other brominated solvents?
A: 2-Bromobutane is stipulated when a secondary alkyl bromide is required for specific organic reactions, such as the synthesis of alkenes or in nucleophilic substitution processes. Its balance of volatility and reactivity makes it advantageous for precise chemical transformations.Q: Where does 2-Bromobutane find its most common use?
A: 2-Bromobutane is most commonly found in research laboratories and manufacturing plants involved in organic and pharmaceutical chemistry, particularly as a reagent in the synthesis of more complex molecules.Q: What is the process for producing 2-Bromobutane at an industrial scale?
A: Industrial production of 2-Bromobutane typically involves the direct bromination of butane or secondary butanol using controlled conditions to ensure high yield and purity. The process is closely monitored for temperature and reaction time to achieve the optimal 98% purity.Q: What are the solubility characteristics of 2-Bromobutane and how do they affect its usage?
A: 2-Bromobutane is insoluble in water but dissolves readily in ethanol and ether. This property makes it suitable for reactions and applications that require non-aqueous solvents, expanding its utility in various synthesis and extraction processes.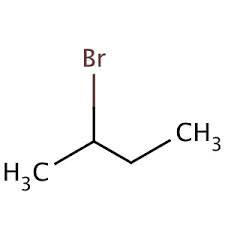

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Aliphatic Bromide Category
Hydrobomic Acid
Minimum Order Quantity : 1 Kilograms
Boiling point : Decomposes before boiling
Usage : Used in glass manufacturing, agriculture, antiseptic, insecticide
Structural Formula : B(OH)3
Molecular Weight : 61.83 g/mol
Smell : Other, Odorless
Lithium Bromide 50-55% Sol
Minimum Order Quantity : 1 Kilograms
Boiling point : ~126.5 C (solution)
Usage : Refrigerant Absorbent, Desiccant, Heat Transfer Medium
Structural Formula : LiBr
Molecular Weight : 86.85 g/mol
Smell : Other, Odorless
N- Butyl Bromide
Minimum Order Quantity : 1 Kilograms
Boiling point : 101.5 C
Usage : Used as alkylating agent, flavor intermediate, laboratory reagent
Structural Formula : CH3(CH2)3Br
Molecular Weight : 137.02 g/mol
Smell : Other, Sweet, Etherlike Odor
N-Pentyl Bromide
Minimum Order Quantity : 1 Kilograms
Boiling point : 130132C
Usage : Intermediate for pharmaceuticals and agrochemicals
Structural Formula : CH3(CH2)4Br
Molecular Weight : 151.05 g/mol
Smell : Other, Characteristic odor

 Send Inquiry
Send Inquiry
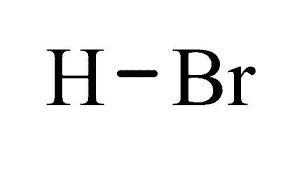
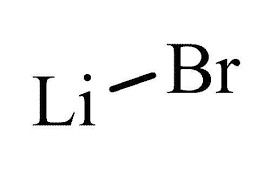
 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese