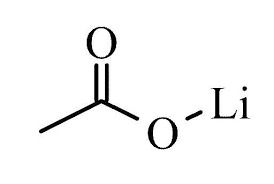
About Lithium Acetate Dihydrate
| Name of Product | Lithium Acetate dihydrate | IUPAC Name | lithium;acetate;dihydrate |
| CAS No | 6108-17-4 | InChI | InChI=1S/C2H4O2.Li.2H2O/c1-2(3)4;;;/h1H3,(H,3,4);;2*1H2/q;+1;;/p-1 |
| Formula | CH3COOLi · 2H2O | InChI Key | IAQLJCYTGRMXMA-UHFFFAOYSA-M |
| Other name / synonyms | LITHIUM ACETATE HYDRATE | Canonical SMILES | [Li+].CC(=O)[O-].O.O |
| LITHIUM ACETATE DIHYDRATE | EC Number | 612-081-5 |
| ACETIC ACID LITHIUM SALT DIHYDRATE | UNII | T460029GB5 |
| Quilonum |
|
|
| Lithium Acetate Anhydrous |
|
|
| MONOCLONAL ANTI-LEF-1/TCF*(TRANSACTIVATI |
|
|
| LITHIUM ACETATE DIHYDRATE, CRYSTALLIZED |
|
|
| LITHIUM ACETATE DIHYDRATE SIGMAULTRA |
|
|
Key PropertiesLithium Acetate Dihydrate is recognized for its high purity, solubility in water and ethanol, and convenient white crystalline appearance. With a melting point of 57C and a density of 1.52 g/cm3, it serves as a dependable reagent in various laboratory applications, particularly in biochemical research and lithium compound synthesis.
Applications in Research and IndustryThis compound is extensively used for DNA and RNA precipitation in molecular biology, buffer preparation, and as a precursor for other lithium-based chemicals. Its reliability and consistency make it an essential choice for laboratory-grade requirements, supporting both academic and industrial research sectors.
FAQ's of Lithium Acetate Dihydrate:
Q: How is Lithium Acetate Dihydrate commonly used in the laboratory?
A: Lithium Acetate Dihydrate is used in biochemical research, notably in DNA and RNA precipitation protocols, buffer preparation, and as a starting material for synthesizing other lithium compounds. Its excellent solubility in water and ethanol enhances its effectiveness in these applications.
Q: What are the benefits of choosing a laboratory grade Lithium Acetate Dihydrate?
A: Opting for laboratory grade Lithium Acetate Dihydrate assures high purity (99%), which is critical for reproducible and accurate results in research experiments. Its reliable performance minimizes contaminants and improves the quality of scientific procedures.
Q: Where is Lithium Acetate Dihydrate sourced or exported from?
A: Lithium Acetate Dihydrate is manufactured, supplied, and exported widely from India, where reputable suppliers ensure stringent quality control and consistent laboratory grade standards to meet international research and industrial demands.
Q: What is the process of manufacturing Lithium Acetate Dihydrate?
A: Lithium Acetate Dihydrate is typically synthesized by reacting high-purity acetic acid with lithium compounds. The resulting product is purified and crystallized, ensuring it reaches the 99% purity required for laboratory-grade reagents.
Q: When should Lithium Acetate Dihydrate be used instead of other lithium salts?
A: Lithium Acetate Dihydrate is particularly recommended when solubility in water or ethanol is crucial, or when a non-chloride lithium source is desired. Its use is common in molecular biology workflows and chemical preparations where purity and minimal interference are priorities.
Q: How should Lithium Acetate Dihydrate be stored and handled?
A: Store Lithium Acetate Dihydrate in a well-sealed container in a cool, dry place away from direct sunlight and moisture. Standard laboratory safety protocols, such as wearing gloves, goggles, and appropriate lab attire, should be followed during handling.
Q: What advantages does Lithium Acetate Dihydrate offer researchers?
A: Researchers benefit from its high solubility, purity, and neutral odor, as well as its proven effectiveness in complex biochemical procedures. Its consistent quality helps ensure reliable data and streamlined laboratory processes.